Automated ESR-Important Facts
Date : 13 Apr, 2018
The gold standard for the determination of the Erythrocyte Sedimentation Rate (ESR) is the Westergren method. Other methods to measure the ESR have become available. They range from modest modifications of the Westergren method to very different methodologies. The ICSH therefore established a Working Group to investigate these new approaches and compile recommendations for their validation and verification. The ICSH (International Committee for the Standardization in Hematology) guidelines for the standardization of ESR, was published in the April 2017 edition.
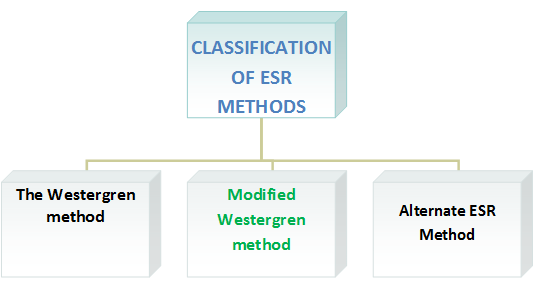
Classification of ESR methods
The Working Group classifies ESR methods into three categories:
Possible advantages of Modified Westergren Method:
- Reduced exposure of laboratory personnel to infectious agents
- Ability to use standard EDTA tubes
- Reduction in analysis time from 1 h to Minutes/seconds
- Reduction in the probability of human error
- Reduction in the amount of labor needed, leading to increased economic efficiency
- Ability to interface instrumentation to the EMR, reducing transcription errors and allowing instantaneous communication of results to clinical staff
- Proficiency Testing Survey Reports indicates that Westergren-based results usually correlate very well with each other
- In addition, it should be mentioned that major advantages of the use of EDTA samples are as follows:
- Avoidance of rejections of many samples in everyday practice
- Reduction in the blood volume required for hematological tests
- Preservation of the red cells morphology, with maintenance of optimal blood stability
- The increased automation reduces the probability of human error and increases economic efficiency
Alternate methods:
Instruments based on non-Westergren/alternative methodology that have not been validated by the manufacturer as outlined below should not be accepted for clinical use. In addition to differences in results, some of the new methods do not measure all the ESR phases (2). Therefore they may show different susceptibilities to interferences. It may be influenced differently by the presence of anemia, or may have different sensitivities and specificities for different disease states eg. paraproteinemia (3) than the traditional or modified Westergren method. There are reports in the literature that patients with hypofibrinogenemia may have a lowered ESR, and patients with afibrinogenemia may have an ESR of zero (4, 5) It is unclear whether the alternative methods will similarly reflect low fibrinogen levels.
Recommendation:
It is the recommendation of New ISCH 2017 Working Group that only the methods validated according to the well-defined criteria (6) should be considered for routine clinical testing. Manufacturers should clearly state whether the results obtained with their instruments can be traced to the Westergren method
User Obligations for modified and alternate ESR methods:
- Perform studies to determine the suitability of the method for their patient population
- Accuracy, Precision, AMR, Carryover, Verifying reference Ranges need to be carried out
- Consider adding an interpretative comment to every result stating that “This result was obtained with an ESR instrument that is not based on the standard Westergren method. The sensitivity and specificity of this method for various disease states may be different from the standard Westergren method”
- Use of commercial ESR QC spanning the analytical range of their instruments is recommended to be run at least once every day
- The laboratory should subscribe to an EQA program specific for its method. If an EQA program suitable for the laboratory’s method is not available, regular (two to three times a year) comparison studies with other laboratories should be performed
Notes:
Transasia is the single distributor across India, for Vesmatic & Vesmatic Cube 80/30 Series Automated ESR analyzers from Diesse Diagnostics, Italy.
Ves Cube series uses standard EDTA tubes; samples are allowed to settle for 20 min, and results are converted to Westergren units. These systems are based on Modified Westergren Method as per ICSH recommendations.
Literature:
- Jou JM, Lewis SM, Briggs C, et al. ICSH review of the measurement of the erythrocyte sedimentation rate. Int J Lab Hematol. 2011;
- Hardeman MR, Levitus M, Pelliccia A, Bouman AA. Test 1 analyser for determination of ESR. 1. Practical evaluation and comparison with the Westergren technique. Scand J Clin Lab Invest. 2010;
- Raijmakers MT, Kuijper PH, Bakkeren DL, Vader HL. The effect of paraproteins on the erythrocyte sedimentation rate: a comparison between the StarrSed and TEST 1. Ann ClinBiochem. 2008;
- Mehta S, Mehta SR, Malhotra H, Sharma UB, Varma AR. Congenital afibrinogenaemia. J Assoc Physicians India. 1989; 37:668‐669.
- Stephan JL, Zeller J, Hubert P, Herbelin C, Dayer JM, Prieur AM. Macrophage activation syndrome and rheumatic disease in childhood: a report of four new cases. ClinExpRheumatol. 1993.
- ICSH recommendations for modified and alternate methods measuring the erythrocyte sedimentation rate April 2017
References:
- Edited from ICSH recommendations for modified and alternate methods measuring the erythrocyte sedimentation rate April 2017
- Technical detail available on Website : -http://www.diesse.it/en/