Fresh Frozen Plasma (FFP) is a blood product made from the liquid portion of whole blood, it is used to treat conditions in which there are low blood clotting factors (INR>1.5) or low levels of other blood proteins.
FFP was used for the first time during the Second World War. It is on the World Health Organization’s (WHO) List of Essential Medicines. FFP is obtained from whole blood or plasma donations via apheresis. Apheresis is a process whereby blood is drawn from a donor and split into its constituent parts, usually the plasma and platelets are retained and the remaining components are returned to the donor. Plasma is rapidly frozen, stored at below −25°C, and rapidly thawed prior to use. It contains a range of coagulation factors. The fibrinogen concentration is 2 to 4 mg/ml. Once thawed, it should be kept at 4°C and should be administered within 24 hours.
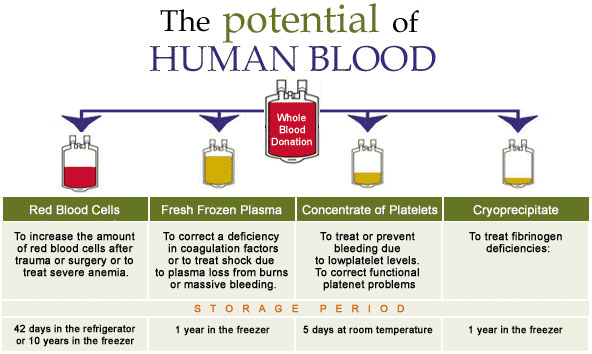
Fresh frozen plasma is used to treat bleeding associated with an increased PT (>1.5 times normal) or aPTT (>2 times normal). In an average-sized adult, one unit of fresh-frozen plasma increases coagulation factors by around 3%. For coagulopathic bleeding, a dose of 10 to 15 ml/kg (typically about 4 units) is sufficient for initial treatment. FFP is not indicated for intravascular volume expansion and is generally overused in cardiac surgery patients, in whom the main hemostatic problem is usually platelet dysfunction.
There are limited specific indications for the use of FFP exists for the treatment of deficiencies of coagulation proteins. Due critical nature of coagulopathies and unavailability of satisfactory alternative therapy, FFP may be appropriate.
Indications for the use of FFP include the following:
- FFP is used to treat rare bleeding disorders like factor V deficiency.
- FFP can be used to reverse the warfarin effect in patients who are undergoing anticoagulant treatment with warfarin, are deficient in the functional vitamin K dependent coagulation factors II, VII, IX, and X, as well as proteins C and S.
- FFP can be used as a antithrombin III supplement in the patients who are undergoing surgery or who require heparin treatment for thrombosis and are deficient of antithrombin III.
- FFP can be used as effective treatment in pediatric patients with secondary immunodeficiencies along with sever protein-losing enteropathy;
- FFP may be beneficial for the treatment of thrombotic thrombocytopenic purpura.
Although FFP is used in the treatment of many severe to chronic illnesses, there are risks involved. The transmission of blood borne diseases, allergic reaction, complication in acute lung injury treatment, and hypovolemia (i.e., a fluid overload and cardiac failure) are few of the prominent risks associated to FFP treatment. The only way out to handle all these risks are to gauge clinical parameters present in FFP. The analysis of blood coagulation factors is commonly performed in the blood banks and clinical laboratories because it plays a central role in providing important information to the physician so as to minimize the patient’s risk. Among these tests, fibrinogen and blood coagulation factor VIII (FVIII) are often analyzed in clinical laboratories to monitor the extent of the introduced anticoagulation.
Cryoprecipitate (Cryo)
In 1950 concentrated antihaemophilic factor or factor VIII was produced from FFP to treat haemophilia. The improvised procedures were developed to produce FVIII concentrate which lead to Cryoprecipitate (Cryo) preparation. Cryo is a frozen blood product obtained from FFP. Cryo was prepared by slow thawing of FFP 1–10°C. FFP is then centrifuged and the precipitate is collected. Cryoprecipitate is enriched with fibrinogen and also high concentrations of FVIII, von Willebrand factor, and factor XIII. Its common use today is to replenish fibrinogen levels during coagulopathies associated with massive haemorrhage, in which fibrinogen decreases to a critical level due to clotting, dilution, blood loss, or all.
Each Cryo unit (approx. 10 to 15 mL) contains; Fibrinogen 150–250 mg (half-life of 100–150 hours), Factor VIII 80–150 U (half-life of 12 hours), von Willebrand factor 100-150 U (half-life of 24 hours), Factor XIII 50–75 U (half-life of 150–300 hours) and fibronectin. Cryo are clinically used in, Haemophilia (Used when factor concentrates are not available), von Willebrand disease, Hypofibrinogenaemia (low fibrinogen levels), Afibrinogenemia, Bleeding from excessive anticoagulation, Massive haemorrhage, Disseminated intravascular coagulation (DIC) and Uremic bleeding tendency.
Coagulation studies
Fibrinogen plays a key role in the blood coagulation cascade, as it can be activated by thrombin and polymerized insoluble fibrin. Meanwhile, coagulation Factor VIII participates in the blood clotting cascade as a cofactor of Factor IXa in the activation of Factor X. The congenital deficiency or abnormality of coagulation FVIII results in a severe bleeding disorder, hemophilia A, which is potentially life-threatening. Low plasma levels of coagulation FVIII leads to a tendency towards clotting inefficiencies, whereas high plasma levels of coagulation FVIII can result in various thrombotic diatheses, such as deep vein thrombosis. Rapid and accurate detection of fibrinogen and FVIII is valuable to evaluate response to therapy and is available for identifying risk control for patients. However, the current laboratory test for fibrinogen and coagulation FVIII is time consuming and expensive. Therefore, a rapid and convenient method for monitoring concentrations of fibrinogen and FVIII is urgent for both physicians and patients. Since the APTT duration was decided by concentration of factor VIII, the concentration of factor VIII can be calculated from the APTT time.
Erba Thrombin Reagent is used in the quantitative determination of fibrinogen in human plasma using the Clauss Method. Erba Factor VIII Deficient Plasma is used for the quantitative determination of the Factor VIII in patients suspected of having a congenital or acquired deficiency of this coagulation protein.
References:
- Bleeding, Hemostasis, and Transfusion Medicine; Jerrold H. Levy, Andrew McKee; Cardiothoracic Critical Care, 2007
- Guidelines for the use of fresh frozen plasma; D. De Backer, B. Vandekerckhove, S. Stanworth, L. Williamson, C. Hermans, P. Van der Linden, R. Hübner, P. Baele, K. Jochmans, A. Ferrant, M.Lambermont, L. Muylle & M. Toungouz; Acta Clinica Belgica, 2008; 63-6
- Guideline for the diagnosis and management of the rare coagulation disorders; Andrew D. Mumford, Sam Ackroyd, Raza Alikhan, Louise Bowles, Pratima Chowdary, John Grainger, Jason Mainwaring, Mary Mathias and Niamh O’Connell; British Journal of Haematology, 2014, 167, 304–326
- Detection of Fibrinogen and Coagulation Factor VIII in Plasma by a Quartz Crystal Microbalance Biosensor; Chunyan Yao, Ling Qu, Weiling; Sensors 2013, 13, 6946-6956
- Cryoprecipitate therapy; B. Nascimento, L. T. Goodnough, J. H. Levy; British Journal of Anaesthesia 2014 Dec; 113(6): 922–934.