INTRODUCTION
The erythrocyte sedimentation rate (ESR) test is one of the most widely performed laboratory tests. ESR is used to assess the acute phase response in inflammation and measures the distance erythrocytes should fall after 1 hr. in millimeters; it is generally affected by size, shape, and number of red blood cells (RBCs), RBC surface charge and aggregation, hematocrite, plasma protein concentrations, especially fibrinogen and temperature. In recent years, novel methods that measure the ESR directly from a standard EDTA-anticoagulated tube have been developed.
Two of these methods are the Ves-Matic Cube 200 (Diesse Diagnostica Senese, Italy) and iSED (Alcor Scientific, USA). The first method, the Ves-Matic Cube 200, uses analog sensors and provides a 20- min ESR result normalized due to the Westergren international standard. The second method, the iSED, uses photometric rheoscope method. Both the methods use lyophilized EDTA, so there is no dilution effect. The tubes are directly put into the analyzers, which reduces the risk of contamination, and also is more practical for routine intense work.
The aim of this study was to evaluate the performances of new devices—the iSED and Ves-Matic Cube 200, and to compare their analytical performance with the reference Westergren method.
MATERIAL and METHODS
Patient Samples
This prospective study included blood samples from 136 patients (87 females and 49 males) selected randomly from both inpatients and outpatients who were admitted to Ankara Numune Education and Research Hospital in June 2013. Citrated blood samples were used for ESR measurements by the Westergren method and K2EDTA-anticoagulated blood samples were used for ESR measurements in the Ves-Matic Cube 200 system and iSED system. Each sample was analyzed by the Westergren method, iSED, and Ves-Matic Cube 200; and the results were compared with the results of the Westergren method.
Blood samples were analyzed within 4 hr. after collection, according to the ICSH recommendations. This assessment was performed according to the recommendation of the ICSH besides the method comparison and ESR reference standards from the Clinical and Laboratory Standards Institute.
The Westergren Method
The conventional manual Westergren method was applied by diluting four volumes of blood with one volume of sodium citrate, according to the ICSH protocol. Citrate-diluted blood was aspirated in Westergren pipette, Westergren-type glass pipettes mounted vertically in a rack. Sedimentation was evaluated visually after 60 min.
The Ves-Matic Cube 200 System
ESR measurement by the Ves-Matic Cube 200 system is based on the modified Westergren sedimentation technique. EDTA-anticoagulated blood samples are homogenized automatically and put into a test tube holder chain. The initial height of the blood column is recorded after scanning directly through the tube by an optoelectronic light source. After 20 min, the final height of the RBC layer is scanned by the optical system again, and the difference is recorded. The results are corrected using the temperature correction equation at 18°C and transformed to 60-min measurement according to the manufacturer’s recommendations. The results are given in millimeter per hour.
The iSED System
The iSED system uses the photometric rheoscope method. The device contains a micro flow cell with the critical kinetics to capture the Rouleaux formation of the RBCs. The system works directly from dry EDTA tube. It is also suitable for pediatric samples using 100μl blood. The iSED device gives the results in 20 sec and the results are expressed as millimeter per hour.
Interference Studies
Ten randomly selected samples were measured by the iSED and Ves-Matic Cube 200 analyzers. Lipemia and hemolysis were performed for interference studies. Lipid solution was used for lipemia interference studies. We used ClinOleicR _ 20% solution (Eczacıbas¸ı, Baxter, Turkey) as lipid solution. Ninety-nine volumes blood and one volume lipid solution were mixed. Hemolysate solution was used for hemolysis interference studies. Hemolysate solution was created by mixing 1,490μl of distilled water and 10μl of blood. Nine volumes blood and one volume hemolysate solution were mixed. Similarly, physiological saline was added to selected ten samples in order to eliminate the effect of dilution and evaluate the net impact of lipemia and hemolysis (99 volumes of the blood, one volume physiological saline for lipemia and nine volumes of the blood, one volume physiological saline for hemolysis). Finally, mixed samples were measured by the iSED and Ves-Matic Cube 200. Treated samples with lipid solution and hemolysate solution were labeled as “Spiked” and untreated samples were identified as “Neat.”
Stability Studies
Twenty samples were chosen randomly and ten of them were stored at room temperature (25°C).Ten of these samples were stored at 4°C and were then allowed to return to room temperature before retesting. ESR measurements were performed by the iSED and Ves-Matic Cube 200 at 4, 6, 8, and 24 hr. after collection.
Precision Study
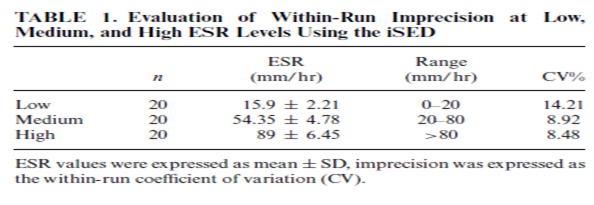
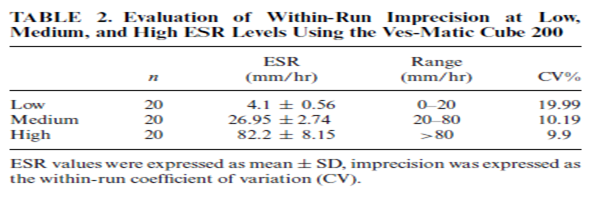
Patients’ samples with low (<20 mm/hr.), medium (20–80 mm/hr.), and high (>80 mm/hr.) levels of ESR were analyzed by 20 replicate measurements in the iSED and Ves-Matic Cube 200. Means, standard deviations, and coefficients of variations were calculated.
Statistical Analysis
The conformity of continuous variables to normal distribution was tested with Kolmogorov–Smirnov test. A paired Student’s t-test and Wilcoxon t-test were used for comparing the means of groups. Values of P < 0.05 were accepted as statistically significant. Passing–Bablok regression analysis was performed. Bias and 95% limits of agreement assessments were performed using Bland–Altman analysis. The nonparametric test of Spearmen’s rank correlation was used to evaluate correlation (r, correlation coefficient). The calculations were performed by using “MedCalc” statistical software (ver. 12.3.0.0) and “The Statistical Package for Social Sciences for Windows”(SPSS v18) software.
RESULTS
Evaluation of Within-Run Imprecision
The within-run imprecisions were 14.21%, 8.92%, and 8.48% for the iSED and 19.99%, 10.19%, and 9.90% for the Ves-Matic Cube 200 for low, medium, and high levels, respectively (Tables 1 and 2).
Evaluation of Stability
The results of the stability studies of the iSED are shown in Table 3.When the specimens are stored at room temperature (25°C), the ESR results did not change significantly after 24-hr collection, but the ESR results of refrigerated (4°C) samples were found to be significantly higher. The results of the stability studies of the Ves-Matic Cube 200 are shown in Table 4. When the specimens are stored at 4°C, the ESR results did not change significantly after 24-hr collection, but the ESR results of the samples at room temperature (25°C) decreased significantly at 6 and 24 hr.
Evaluation of Interference
There was a significant positive correlation between C-reactive protein (CRP) levels and ESR results measured by the Ves-Matic Cube 200 (r =0.654, P < 0.001). RBC count and hematocrit levels were negatively correlated with ESR results measured by the Ves-Matic Cube 200 (r=−0.597, P < 0.001; r=−0.615, P < 0.001, respectively). There was a significant positive correlation between CRP levels and ESR results measured by the iSED (r = 0.643, P < 0.001). RBC count and hematocrit levels were negatively correlated with ESR results measured by the iSED (r=−0.299, P = 0.001; r=−0.441, P < 0.001, respectively). The presence of lipids appears to cause a falsely low ESR measurement in the iSED (P=0.005) and Ves-Matic Cube 200 (P = 0.007). Similarly, hemolysis does cause a falsely low ESR measurement in Ves-Matic Cube 200 (P = 0.005). On the contrary, hemolysis does not cause significant difference in the measurement of ESR in iSED device (P = 0.766; Tables 5 and 6).
Method Comparison Study
ESR results measured by using the iSED and Ves-Matic Cube 200 analyzers were compared with the reference Westergren method. ESR measurements in 136 samples resulted in a mean bias ESR of 1.4 mm/hr. (95% confidence interval [CI], −34.4 to 37.2) using the Ves MaticCube-200 and a mean bias ESR of 13 mm/hr. (95% CI,−35.7 to 61.6) using the iSED (see Figs. 1 and 2). Passing–Bablok regression analysis yielded the equation “y = 0.92x + 1.25” between the Ves-Matic Cube 200 between the iSED method and Westergren method. The iSED method (n = 136) yielded a slope of 0.74 (95% CI, 0.61–0.84) with an intercept of 0.07 (95% CI, −2.32 to 1.89; Fig. 4). The iSED sedimentation method showed a poor correlation with the Westergren method (r = 0.76, P < 0.0001).

According to Bland–Altman analysis, this resulted in a mean bias of 13 (95% limit of agreement between 61.6 and −35.7). The Ves-Matic Cube 200 method showed a moderate correlation with the Westergren method (r =0.84, P < 0.0001). According to Bland–Altman analysis, this resulted in a mean bias of 1.4 (95% limit of agreement between 37.2 and −34.4). We divided the results (n = 136) into subgroups according to the Westergren ESR levels as <20, 20–80, >80mm/hr (low, medium, high levels, respectively). Bland–Altman analysis, Passing–Bablok regression analysis, and Spearman’s rank correlation analysis were performed separately for these levels. The results of Bland–Altman, Passing–Bablok, and Spearman’s rank correlation analysis are shown in Tables 7–9, respectively.
The iSED sedimentation method showed poorer correlation with the Westergren method in increscent ESR levels (low levels r = 0.67, medium levels r = 0.48, high levels r = 0.27).
DISCUSSION
The ESR test, described about 70 years ago, is one of the most widely performed laboratory tests. The Westergren method was recommended to measure the ESR by the International Council (previously Committee) for Standardization in Haematology in 1973.






Although traditional Westergren method is accepted as the gold standard method for the measurement of ESR, but being time-consuming, laborious, and having contamination risks it is not preferred for routine use. There is a need to establish newer systems in order to overcome these problems. Several automated systems are now available for ESR measurements. The Ves-Matic Cube 200 and iSED are analyzers that use new methods for ESR measurements. These systems were shown to provide faster, easier, more practical, and biologically safer ESR measurements.
One of these methods is used by the Ves-Matic Cube 200 analyzer, has an optical system through which light passes and gives results in 20 min, and also the temperature correction settings are available. Another method, photometric rheoscope, is used by the iSED analyzer, gives results in 20 sec, and there are no temperature correction settings. The current study was planned to evaluate the analytic performance of these analyzers by comparing it with the Westergren method. According to CLSI H2-A4 guideline, acceptable performance limits are defined for different ESR concentrations and CVs% (where CV is coefficient of variation) calculated from acceptable performance limits are between10.88 and 38.88 for different ESR values. CV% values in low ESR concentrations increase when compared with CV% values in high ESR values. In our study, we studied patient samples with low (<20 mm/hr.), medium (20–80 mm/hr.), and high (>80 mm/hr.) levels of ESR with 20 replicate measurements.
The within-run imprecisions were 14.2%, 8.92%, and 8.48% for the iSED and 19.9%, 10.1%, and 9.90% for the Ves-Matic Cube 200 in low, medium, and high levels, respectively. Both the iSED and Ves-Matic Cube 200 seem to have precisions within the acceptable performance limits given in CLSI H2-A4 guideline Curvers et al. studied with 244 patients and reported CVs for the Ves-Matic Cube 200 analyzers as 19.6%, 10.9%, and 5.6%. Perovic et al. reported CVs for Ves-Matic Cube 200 analyzers as 9.19%, 13.88%, and 5.66% (9). Sezer et al. had evaluated the Ves-Matic Cube 200 system and reported CVs as 14.01%, 14.99%, and 5.69% for low, medium, and high ESR levels, respectively. Happe et al. evaluated the precision for Diesse Mini-Ves (DMV) and reported that precision decreased substantially in low ESR levels (CVs: 30.4%; (10)). These findings are supported by our results. To our knowledge, there has been no report including the iSED imprecision study.
ESR results of the iSED, when the specimens are stored at room temperature, did not change significantly after 24-hr collection but ESR results of refrigerated samples increased significantly. We believed that the increase in the refrigerated samples was dependent on the reoscopic method based on the viscosity (depend on viscosity). The Ves-Matic Cube 200 ESR results, when the specimens are stored at refrigerator, did not change significantly after 24-hr collection but ESR results of room temperature samples decreased significantly at 6 and 24 hr. The decrease in samples after 24 hr has been supported by other researchers. They reported that this can depend on swelling of erythrocytes and decrease in the sialic acid of the erythrocytes membranes. Perovic et al. investigated the stability for 24-hr storage at 4°C or room temperature and reported that the highest differences were obtained after 24-hr storage of the samples at room temperature. Sezer et al. performed the Ves-Matic Cube 200 stability study on 101 samples. Similar to our findings, they found that ESR results decreased significantly in specimens stored at room temperature for 24 hr, but did not reported a change in refrigerated samples.
Lipemia caused significantly low ESR measurements in both the iSED (P=0.005) and Ves-Matic Cube 200 methods (P = 0.007). Hemolysis caused a significant decrease in ESR levels for the Ves-Matic Cube 200 method (P =0.005), though there has not been a significant difference in the measurement of ESR for the iSED method (P =0.766) with hemolysis. Sezer et al. found that presence of lipids and hemolysis caused falsely low ESR measurements in the Ves-Matic Cube 200 analyzer and that was in accordance with our findings. For the Ves-Matic Cube 200, systematic bias was calculated as 1.4 and limits of agreement were found in the range of−34.4 to+37.2. Systematic bias for the Ves-Matic Cube 200 was found to be higher in increscent ESR levels (mean bias of low levels = −6.2, mean bias of medium levels = 0.6, mean bias of high levels = 16.7). Sezer et al. reported systemic bias as −0.7 and limits of agreement as −32.6 to 31.2; Perovic reported the bias as −0.5, limits of agreement −13.9 to 12.9 in the Ves-Matic Cube 200 analyzer and Westergren method comparison study.
There has not been a method comparison study including the iSED analyzer. In our study, systematic bias was calculated as 13 and limits of agreement were found in the range of −35.7 and +61.6. For the iSED, systematic bias was found higher in increscent ESR levels (mean bias of low levels=0.4, mean bias of medium levels=13.8, mean bias of high levels = 32.6). In their study Perovic et al. evaluated the accuracy that was established using a 95% CI for the mean difference between the Ves-Matic Cube 200 and Westergren method (mean of difference: 0.47 ± 6.84 mm/hr; 95% CI, −0.376 to 1.325; (9)). Correlation coefficient was 0.946 (P < 0.001). The Ves-Matic Cube 200 showed a good correlation with the Westergren method. Happe et al. evaluated the performance of DMV. They showed a close correlation between the Westergren and four-sample DMV ESR methods (r =0.966; P < 0.001;They suggested the use of four-sample DMV ESR analyzer instead of the Westergren method for ESR measurements in hospitals or clinical laboratories.
Caswell et al. showed good agreement with the Ves-Matic system and ICSH values with a tendency to false high results. The high results may be due to loss of vacuum during storage of tubes and loss of water vapor through the plastic or absorption of citrate. But we did not face this problem because we used dry EDTA. According to our results (Table 9), a poorer correlation between the Ves-Matic Cube 200 method with the Westergren method (r = 0.84) was observed, though the correlation was better when compared with iSED analyzer, which had a correlation coefficient of 0.76. The iSED and Ves-Matic Cube 200 sedimentation method showed poorer correlation with the Westergren method in increscent ESR levels. Our findings were in accordance with Curvers et al. study that reported the correlation coefficient as r = 0.83 (95% CI, 0.76–0.88) for the Ves-Matic Cube 200 analyzer. We found a negative correlation between ESR results of the Ves-Matic Cube 200 and iSED with hematocrit levels (r = −0.615, r = −0.441, respectively; P < 0.0001, P < 0.0001, respectively).RBC count (r=−0.597, r=−0,299, respectively; P < 0.0001, P = 0.0001, respectively) and CRP levels (r = 0.654, r = 0.643, respectively, P < 0.0001, P < 0.0001, respectively) were positively correlated with ESR results of the Ves-Matic Cube 200 and iSED. Sezer et al. showed similar results for the Ves-Matic Cube 200.

As a result, the iSED analyzer cannot be used interchangeably with the Westergren method because of its poor correlation and high bias. The Ves-Matic Cube 200 showed a better but moderate correlation with the Westergren method and had a low bias. Although the systems using EDTA samples have significant advantages, there seems to be a need for the standardization of these systems for ESR measurements.
CONFLICT OF INTEREST
The authors do not have any conflict of interest
REFERENCES
- Edited from original article: Comparison of iSED and Ves-Matic Cube 200 Erythrocyte Sedimentation Rate Measurements With Westergren Method - Nihal Bo˘gdaycio ˘ glu,1 Fatma Meric Yilmaz,1,2∗ Sevilay Sezer,1 and Esra O˘guz - Clinical Biochemistry Laboratory, Ankara Numune Education and Research Hospital, Ankara, Turkey - Clinical Biochemistry Department, Yıldırım Beyazıt University Medical Faculty, Ankara, Turkey - Journal of Clinical Laboratory Analysis 29: 397–404 (2015)
- Curvers J, Kooren J, Laan M, et al. Evaluation of the Ves-Matic Cube 200 erythrocyte sedimentation method: Comparison with Westergren-based methods. Am J Clin Pathol 2010;134:653–660.
- Mahlangu JN, Davids M. Three-way comparison of methods for the measurement of the erythrocyte sedimentation rate. J Clin Lab Anal 2008;22:346–352. J. Clin. Lab. Anal.
- Horsti J,RontuR,Collings A.A comparison between the StaRRsed Auto-Compact Erythrocyte Sedimentation Rate Instrument and the Westergren method. J Clin Med Res 2010;2:261–265.
- Sezer S, Yilmaz FM, Kaya O, Uysal S. Evaluation of Ves-Matic Cube 200 for erythrocyte sedimentation rate determination. J Clin Lab Anal 2013;27:367–372.
- ICSH Recommendations for Measurement of Erythrocyte Sedimentation Rate. International Council for Standardization in Haematology (Expert Panel on Blood Rheology). J Clin Pathol 1993;46:198–203.
- Caswell M, Stuart J. Assessment of Diesse Ves-Matic automated system for measuring erythrocyte sedimentation rate. J Clin Pathol 1991;44:946–949.
- Koepke JA, Bull BS, Simson E, et al. Reference and Selected Procedure for the Erythrocyte Sedimentation Rate (ESR) Test; Approved Standard—Fourth Edition. NCCLS Document H2-A4. 2000. Vol. 20, p 1–24.
- Krouwer JS TD, Garber CC, et al. NCCLS Method Comparison and Bias Estimation Using Patient Samples; Approved Guideline, 2nd edition. Document EP9-A2. 2002. Vol. 22, p 1–55.
- Perovic E, Bakovic L, Valcic A. Evaluation of Ves-Matic Cube 200—An automated system for the measurement of the erythrocyte sedimentation rate. Int J Lab Hematol 2010;32:88–94.
- Happe MR, Battafarano DF, Dooley DP, et al. Validation of the Diesse Mini-Ves erythrocyte sedimentation rate (ESR) analyzer using the Westergren ESR method in patients with systemic inflammatory conditions. Am J Clin Pathol 2002;118:14–17.
- Lugton RA. The influence of sialic acid on the ESR. Med Lab Sci 1989;46:33–38.